How MGS Powered the Next Generation of GLP-1 Packaging
In the high-stakes world of healthcare packaging, time is the toughest competitor. To meet unpredictable changes in demand for life-changing products, manufacturers must find the perfect balance between massive scalability, rigid compliance, and uncompromising quality.
That’s why when a global CDMO faced skyrocketing demand for GLP-1 based therapies, they turned to MGS for a solution.
The project required the development of a new packaging line spanning tray unloading, labeling, cartoning, case packing, and more, while meeting strict pharmaceutical regulations and achieving high production efficiency. MGS responded with a cost-effective system that brought together tailored engineering, integrated automation, and a cohesive approach to line design.
Scaling production for a surge in demand
The partner is a globally recognized pharmaceutical co-packer supporting major pharmaceutical brands. In this case, the CDMO was newly responsible for packaging a GLP-1 medication administered through prefilled injection pens. As prescriptions for the medication increased worldwide, the CDMO needed to rapidly expand packaging capacity to keep up with the needs of its pharmaceutical client.
However, when the organization began exploring solutions, it found a major challenge in building such a specialized high-speed production line capable of handling injection pens, accessories, and pharmaceutical inserts while meeting strict regulatory standards.
Supporting a global pharmaceutical packaging partner
The sudden market expansion placed significant pressure on the supply chain.
Scaling for such a surge meant engineering a high-speed line from the ground up to reach a minimum of 220-carton-per-minute output. The complexity of the project lay in merging multiple third-party systems into a streamlined design while maintaining the rigorous compliance standards of the pharmaceutical industry. By prioritizing both integration and cost-optimization, MGS transformed a significant supply chain bottleneck into a benchmark for production excellence.
Why MGS was selected
After evaluating several suppliers, the CDMO selected MGS as its integration partner for the project.
What factors gave MGS the advantage:
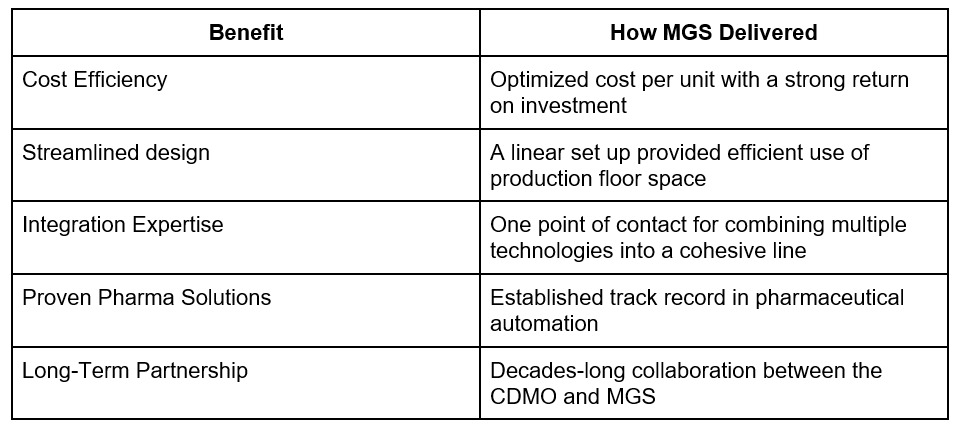
The existing relationship played a particularly important role. Having successfully worked together on multiple projects in the past, the partner trusted MGS to deliver a reliable and scalable solution.
Equally important was MGS’s complete line integration capability, a critical factor for a complex project involving multiple third-party systems.
A cohesive solution for a complex project
The project began with a close collaboration between MGS and the CDMO’s engineering teams to design an integrated solution specifically tailored to meet the site’s unique production and regulatory needs. The process required packaging multiple components, including the pens and literature inserted into a single kit.
To manage this complexity, MGS implemented a series of core system components designed for high-speed kitting, cartoning, and case packing production:
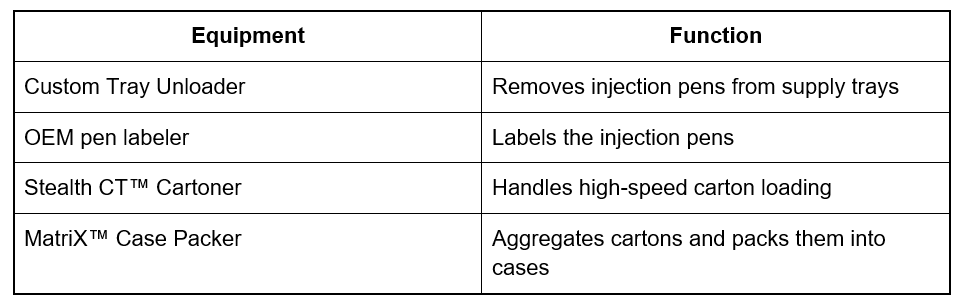
The packaging process begins as injection pens arrive in trays supplied by a third-party manufacturer. MGS engineered a custom tray unloader, using robotic handling to automatically remove the pens and feed them directly into the packaging line.
Then an integrated OEM pen labeler applies a label on each injection pen. Throughout this stage, inspection systems ensure that only compliant products continue through the line to the kitting stage, where the remaining components are assembled.
The prepared kits are then loaded into cartons using the Stealth CT™ cartoner. Key features of this stage include horizontal loading, high-speed operation, and integrated quality inspection and verification: each carton passes over a checkweigher while serialization software prints lot numbers, expiration dates, and unique codes to enable full traceability.
After serialization, cartons move to the final stage of the packaging process. Using the MatriX™ case packer, cartons are automatically grouped into predefined pack patterns and loaded into shipping cases. The system aggregates cartons, linking individual serial numbers to cases to maintain end-to-end traceability.
The project’s impact
By integrating labeling, inspection, serialization and aggregation technologies, MGS created a fully coordinated packaging line.
Key performance advantages:
● 220 cartons per minute production capacity
● Fully integrated serialization and aggregation
● High-accuracy commodity and product inspection and verification
● Optimized cost per unit
Through a combination of custom engineering, proven technology and seamless integration, MGS enabled its partner to meet rising market demand while maintaining the efficiency, compliance and reliability required in modern pharmaceutical manufacturing.
Want to discover how MGS can scale your pharma production? Contact our team today!
